For the implementation of our project in the real world, we have taken many aspects into consideration carefully.
End users-- Who are our proposed end users?
According to our survey and searched data, young adults aged 13-19 years old exhibit vigorous amounts of secretion of oils and fats during puberty. At the same time, young people have insufficient awareness and adapts insufficient precautionary measures towards acne. Not doing good face cleaning will result in oil plugging the pores, which will then trigger the large proliferation of P. acnes. Up to 85% of young people are troubled by acne, which is the main acne pathogenesis population and often has a longer onset time, usually more than one year, mostly 2-4 years. In addition, acne is difficult to heal, which greatly leads to facial appearance anxieties in adolescence, producing emotional uneasiness and threatening their physical and mental health and learning lives.
Considering the economic capacity of adolescents, in order to reduce the price of the products as much as possible while guaranteeing the therapeutic effect of the product, we used a method, lyophilization, that is low-cost but can maintain the activity of our engineered bacteria as much as possible. Freeze and dry product to its moisture at a low temperature, and then put it in a vacuum environment to dry it to let the moisture directly from the solid-state sublimation to water vapor precludes from the article, allowing the article to dry while maintaining the activity of bacteriophages, proteins, etc..
Product-- How do we envision others using our project?
Concerning the form of lyophilized powder of engineered bacteria, we envisioned a process of production and use of our product:
1.Freeze and dry our article using a lyophilizer to make the product
2.Store and transport the
product
After the users mix the lyophilized powder in the product and the medium solution we provide, it can be applied to the face for using after a certain degree of incubation at a suitable temperature. In view of the good therapeutic effect of our products on mild to moderate acne, it can effectively alleviate people’s acne symptoms, and they will no longer be troubled by long-term acne, which greatly reduces the appearance anxiety of people, especially teenagers, and makes them glow with self-confidence. At the same time, it won’t cause drug resistance. The product is named Acneraser, which means an eraser that can remove acne.

As we continue to advance our project in the future, we will also work to measure activation efficiency, seek the most appropriate conditions to ensure the best effect for users.
Commercialization--How would you implement our project in the real world?
Patent protection
When we were engaged in an online communication with Mr. Dajun Yang, chairman of the Ascentage pharma, he reminded us to be sure of invention patent protection for our rare idea before the product was introduced to the market, and can choose to go through a certain stage of research and development after determining a series of conditions, processes of product production.
Every time we communicate with experts and partners, we keep the details of our experiments as much as possible to protect our intellectual property rights. While in popular science education, as well as in communication with stakeholders, we merely introduce the idea of the project’s design. In this process, the team always puts the issue of intellectual property protection at the top to guarantee possibilities for future extension of the project.
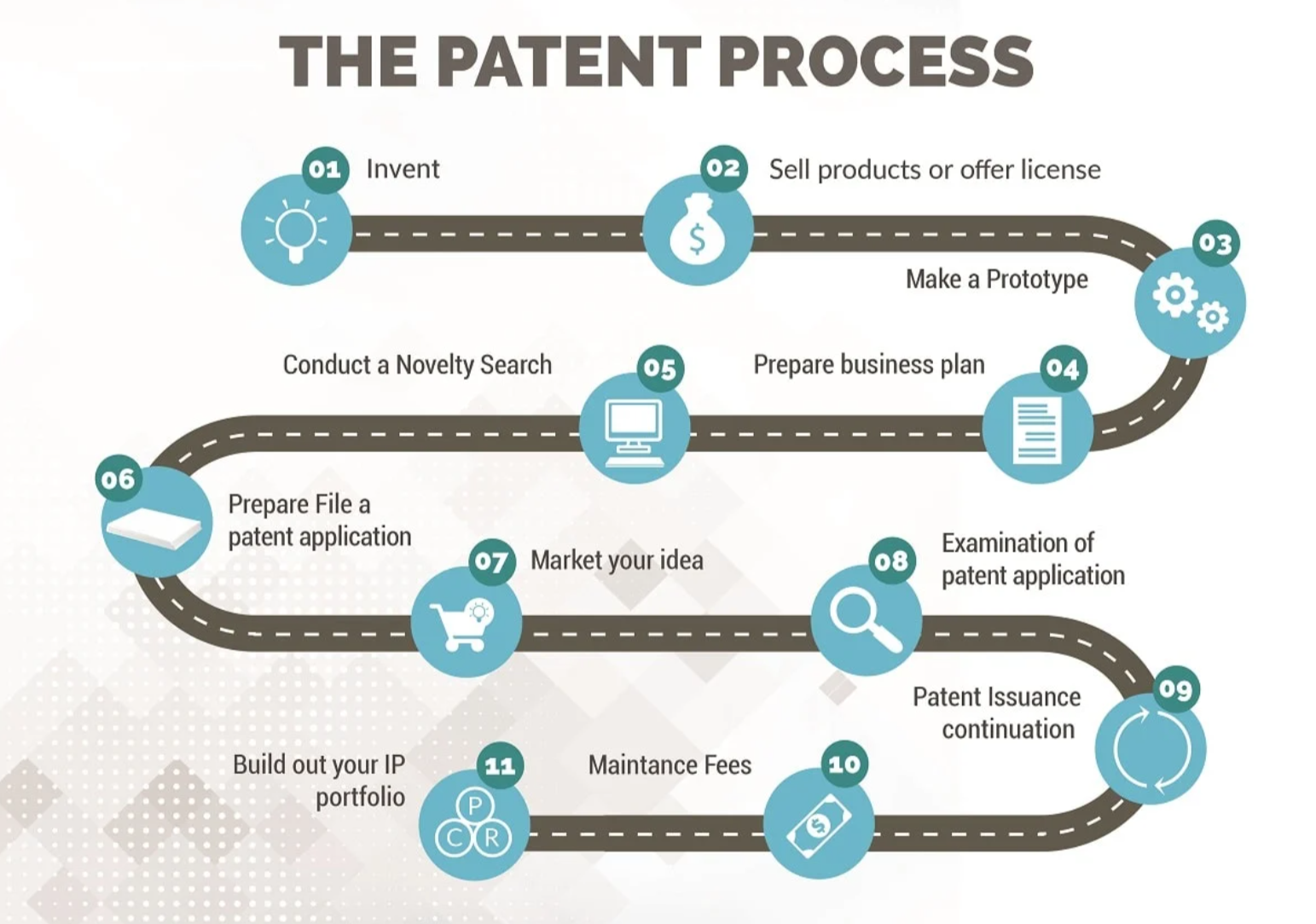
According to the patent law, the application of an invention patent includes five stages: filing, initial trial, publication, real trial, and authorization, and the review flow diagram is as follows:
Profit model
We mainly have three models of profitability:
1.Carry out technical services, help related enterprises to carry out synthetic biology or acne
treatment-related research and development.
2.Sell part of the technology platform, or a certain
technology or patents to relevant research departments to help it develop related products more
quickly and effectively, promoting the development of the whole market environment.
3.Sell
products through collaboration between pharmaceutical representatives, hospitals, pharmacies and
Internet platforms.

In all, in terms of implementing our project in the real world, we have to cooperate with prominent enterprises that have some social influence and marketing means for promoting sales, and well-known trademark brands and mature marketing models are excellent conditions to show the public the entirely new mechanistic product. We have shared the high-level display with Ascentage Pharma and Humanwell Healthcare about our project. Mr. Da Jun Yang, chairman of Ascentage Pharma, said our project was well thought and had great potential to consider issues of good patent protection in advance during the process of continuing R & D sales. He looked forward to seeing our project continuing.
Safety-- What are the safety aspects you would need to consider?
To develop a clinically potent therapy as well as a commercially feasible product, safety and security should be the most important factors to consider. We have recapitulated the biosafety concerns and our corresponding solutions.
Safe and secure lab work
Our team was in full compliance with the safety and security rules and received a safety training. We have a charge teacher and a teaching assistan.
In our experiments, we strictly divided the experimental areas to avoid mutual pollution and carried out our experiment in strict accordance with the experimental operating procedures.
For more details https://2021.igem.org/Team:WHU-China/Safety
Safe and secure project design
When designing our project, we always kept safety concern in mind. The bacteriocin we used specially targets Propionibacterium bacteria, including Propionibacterium acnes. It is safe to our chassis and won’t causing too much influence on skin flora. In addition, we designed fatty acids sensing module to take control of the expression of bacteriocin PctA exactly in our skin, maintaining a balanced, symbiotic relationship between the skin and skin microbiota. Moreover, to avoid environmental pollution caused by engineering bacteria, we constructed a glycine deficient strain which is unable to grow or reproduce in the wild without offering additional glycine.
In the laboratory, we have designed a detailed experimental scheme. Most of the organisms used in our experiments will not pose risks to people, except for Propionibacterium acnes. In the process of the experiment, we avoided direct contact with Propionibacterium acnes.
In future applications, we'll use Escherichia coli Nissle 1917 (EcN) as chassis. Escherichia coli Nissle 1917 (EcN) is a nonpathogenic E. coli strain and one of the best examined probiotic strains. The safety, tolerability, and efficacy of consuming EcN for curing chronic diseases have also been evaluated.
For more details https://2021.igem.org/Team:WHU-China/Design
Safe and secure transfers
In the laboratory, we had the most of genes synthesized and shipped by Tsingke Biotechnology Co., Ltd., who guaranteed safe and secure transfers.
In future applications, we will use a method, lyophilization, and lyophilized powder will be easy and safe to transfer.
Challenges-- What other challenges would you need to consider?
Rivals
When it comes to commercially available acne therapeutics, based on our questionnaire results, adolescents who are the target users of our products are most familiar with acne therapeutics such as erythromycin ointment, adapalene gel, and tretinoin cream, which have been marketed for many years and account for the majority of acne cases market share. Thus, with the help of HFMD, we conducted a Boston matrix analysis of sales share data in the recent years from the retail end of anti-acne products and drew the following conclusions:
- Most of these drugs were star products, with a higher market share and growth rate, and a higher market competition status. They belonged to the main competitors.
- These drugs are all used as acne treatment drugs, but they are almost the same when it comes to the mechanisms.
- On the whole, except for a small number of star products, the vast majority of products are in the lower left and right quadrants, belonging to Taurus products or thin dog products. Taurus products are products with a low growth rate and high market share. A large number of Taurus products in the field represent a mature market, generally belonging to the cash source of enterprises in enterprises, and no longer pay attention to the expansion of market scale; Thin dog products refer to products with low growth rate and low market share. They are products that should adopt a withdrawal strategy. Many such products in acne preparations represent market entry difficulties and barriers.


Public Cognition
The results of the questionnaire survey on adolescents showed that the vast majority of young people has little or no knowledge about live biotherapeutics. Moreover, when we discussed our project with experts from Wuhan Jun Wei An Life Technology Company and HUMANWELL HEALTHCARE, they all indicated that the current biologically based live biotherapeutics were not popular commercially nor were used with traditional chemotherapy of choice.

Our products, unlike traditional chemotherapeutics, are completely new biotherapeutics, meaning that, at the early stage of product marketing, the public has difficulty accepting and trying novel live bacterial products and will have relatively large concerns when selecting them, leading to difficulties in sales of our products. Later on, before the products go out of the laboratory, refine R & D, and produce sales, we need to promote through traditional advocacy models or by means of various platforms on the Internet as well as conducting a series of related popular science or lecture campaigns. This is also shown in our HPS. In addition, we hold the firm belief that as synthetic biology advances, with the help of many companies and organizations, awareness and acceptance will definitely increase.
Reference
[1] http://www.lisbonlx.com/















