HUMAN PRACTICES

Introduction
Amebiasis every year directly affects at least 50 million people, while only 10 percent of them have symptomatic form. Keeping in mind the biopsychosocial paradigm of health [1], each disease has its roots not only in biological but in social, psychological, and environmental dimensions. Therefore, it is important to understand the context of this infectious disease spread and raise awareness about our solutions and prevention. Understanding social, economic, political, cultural behaviors and aspects related to the disease enables the implementation of new external insights to the product design. This way, we can customize our prototypes for them to function at their best and meet end-users needs.
Therefore, analysis via social and moral aspects for us was necessary. As amebiasis is more common in places outside our cultural cognition, internationality were of utmost importance for our integrated human practices. Last but not least, environmental impact is important for us because the world is experiencing a ecological problems. As responsible citizens, we feel the duty to contribute to a greener and more sustainable future.
To follow these values as sincerely as possible, we:
- Analyzed scientific literature about amebiasis’ effect on society to better understand the context of this deadly infection’s emergence and spread.
- Consulted social scientists to better understand social situation in target countries.
- Consulted specialists from the most affected regions, as there was a deficiency of publicly available literature about the ongoing practices in the tropical regions and their effectiveness. We chose affected regions by researching [2, 3, 4, 5, 6] the prevalence of the disease. Those were India, South Africa (Vhembe region), Mexico, and India.
Thought process on Responsible Research and Innovation framework
Using Responsible Research and Innovation framework
We have chosen to use the Responsible research and innovation framework (RRI) [7] for our human practices to be thoughtful and insights to be closely related to the project. Responsible Research and Innovation can be defined as a process by which society and innovators are both accountable for the innovation process and its results to be ethical, sustainable, and desired by the users - society. This way, scientific and technological progress can be appropriately embedded into our society and facilitated for further development [8].
The initial preference for this particular framework evolved by getting familiarized ourselves with the Integrated Human Practices concept in the iGEM context and reading Human Practices activities of previous teams. For each of the meetings, four dimensions oblige to thoroughly reflect on the meeting itself and point out newly gained insights. This research strategy really helps to maintain the focus on the problem, consciously actualize required changes and implement them in the project. While seeking to acquire more evidence-based arguments to arrive at a decision, we have discovered some significant advantages of RRI.
- RRI requires responsive collaboration between science and innovation. It should aim to be sustainable by including stakeholders in the decision-making process [9]. This helps us to more confidently address The United Nations Sustainable Development Goal 17 - partnerships for the goals (you can take a look at how we succeeded here).
- RRI enables us to consider the responsibilities both at the local and global levels [7]. As amebiasis is a global problem, this property of the framework was very beneficial for us.
- RRI has a strong social consideration and promotes relationships with stakeholders, which can lead to desired results of innovations in the social security dimension [10]. This is important for us as we try to tackle an infectious disease, and health impairments also have social and economic impacts.
RRI itself is explained in greater detail on Exeter 2019 team page and in this reference [7]. We mainly followed the four dimension structure and applied it to not only the project as a whole but also to each of the consultations.
Workflow
Our (Integrated) Human Practices workflow over the iGEM cycle looked like this:
- Research about the environmental, socioeconomic, cultural context of the disease and its control measures. At the same time aiming for a proper framework that would enable concise further societal dimension implementation into the project.
- Stakeholder identification and cooperation with stakeholders. Reflection of new insights, selection of the most fundamental ones, and plan of their implementation - an iterative process repeated after each of the consultations.
- Check-point overview of the current status in July and in September. We reviewed the materials we have gained and how they reflect the Responsible Research and Innovation framework. Decided on what other actions to take to gain more insights and followed how gained insights were being implemented.
- Overviewing and evaluating results, organizing information for this page.
Researching the context with values in mind

Responsibility
Even though our synthetic biology project’s routine execution is confined by the walls of the lab, we understand that the end result will affect society. Therefore, we take the responsibility to create not only safe and harmless solutions for amebiasis but also relevant and good for society. In order to do that, we consciously care about the roots of amebiasis, the current state, and the impact that our project would bring. We thoroughly considered such aspects and they are described in Entrepreneurship, Sustainable development impact pages. When the fundamental aspects are considered, it is our responsibility to respond to external insights (research, stakeholders), whether it is modifying test design or creating an educational poster.
By acting and creating, we must not only consider the trail behind amebiasis, diagnostic tests, and probiotics. We understand that we are working in a field that has yet too many controversies - societies still discard GMOs. Therefore it is our duty to communicate and act thoughtfully and ethically, as we are local representatives of synthetic biology.
Interculturality
There were many examples when the help from the global north was not implemented and wasted away because humanitarian help organizers did not understand the different context. This issue was raised by one of our consultants Ieva Giedraityte. This consultation inspired us to empathize with focus cultures and consider differences from our own country.
Some of the reasons for high amebiasis prevalence hides in the cultural peculiarities. Scientists have observed that food handlers in some African regions lack knowledge of appropriate food processing, temperature for food storage and do not have an adequate understanding of sources of either contamination or cross-contamination [11]. Street food is also prevalent where the spread of pathogens is even more supported. Keeping in mind that amebiasis is a foodborne disease, this peculiarity is advantageous for the further spread of the disease. This strengthens the idea for high demand of “AmeBye” tools for natives from high prevalence regions.
Traveling and globalization is another aspect that highly contributes to the spread of E. histolytica. One of the amebiasis symptoms - diarrhea - is the second most common reason why travelers seek medical attention [12]. In one study, a total of 25,867 returned travelers were analyzed, and 12,5 percent of travelers that manifested infectious gastrointestinal disease have been diagnosed with amebiasis [13]. Moreover, immigrants are a well recognized risk group for developing and spreading amebiasis [14], and their position as refugees are even more vulnerable. They often come from hazardous backgrounds, weakened. Until the procedure of granting asylum is finished, they have to live in questionable living conditions, which can add to the progression of the symptomatic or even invasive form of the disease. Such understanding motivates to consider lower prevalence countries as a target group.
Socioeconomic aspect
Infectious disease prevalence is highly associated with the economic status and the development of the regions. This is proved by the epidemiological transition [15] concept - with the development of the country, the progression of its economic and social state occurs. This means an increase in the quality of healthcare together with a decrease in the number of cases of infectious diseases.
Studies show that poor living conditions, previous history of infection in a family member, and an unhygienic toilet facility are significant factors for developing amebiasis [16]. Dr. I. Giedraityte has also emphasized that in the developing regions, there are many people with poor education. Therefore there will be a challenge in reaching our target audience and communicating with them.
With “AmeBye”, we expect an improvement in socio-economic dimensions of equality and individual quality of life as well. With probiotics, a person could avoid infection, and a low-cost, user-friendly diagnostic test is suitable for in-home usage.
Environmental
We are all witnessing climate change, and at every step, we must ensure that our activities are sustainable and good for the environment. Climate change can also impact the prevalence of amebiasis. Studies show that temperature and precipitation are associated with a higher risk of E. histolytica infection. This can be explained by E. histolytica being a water and foodborne parasite. Thus, rainfall could provide favorable environmental conditions for its reproduction and survival [17]. To reduce our environmental impact, we designed our test so it would be easy to disassemble and recycle. We considered the peculiarities of tropical regions and adapted the test accordingly - it can resist high temperatures, humidity, direct sunlight. More information is in our Design page.
Stakeholder identification and communication
Stakeholders
Being students and examining a topic that we had no previous experience with, we knew right away that additional external guidance will be necessary for further development of the project. Therefore we defined our key stakeholders - specialists and institutions that would be affected by “AmeBye” or people whose involvement in the execution of the project could be beneficial. We defined stakeholders by answering such questions:
- Who may be affected negatively (positively) by the successful execution of “AmeBye”? Tropical regions inhabitants, healthcare institutions globally, travelers.
- Who may be affected directly or indirectly? Directly: locals, healthcare institutions, travelers. Indirectly: travel agencies, governments (the reduction in disease scale will leave more people healthy, creating and working)
- Who has the power over the decision-making regarding the project’s development approval, continuation? Healthcare ministry, The State Medicines Control Agency, National Bioethics Committee.
- Who may bring beneficial knowledge to the execution of the project? Scientists working with proteins, diagnostic tests, genome editing, medicines; researches of tropical regions for the contextual information; local leaders for better understanding of the community.
We have created a plan in which we divided all of the stakeholders into the following categories:

We then expanded each of the categories with as many alternative stakeholders as possible. We then organized our list timewise according to the relevance of the consultant to the developed stage of the project. Stakeholder identification was not a one-time task, we continuously complemented the list and brainstormed for new ones.
Documentation of the meetings
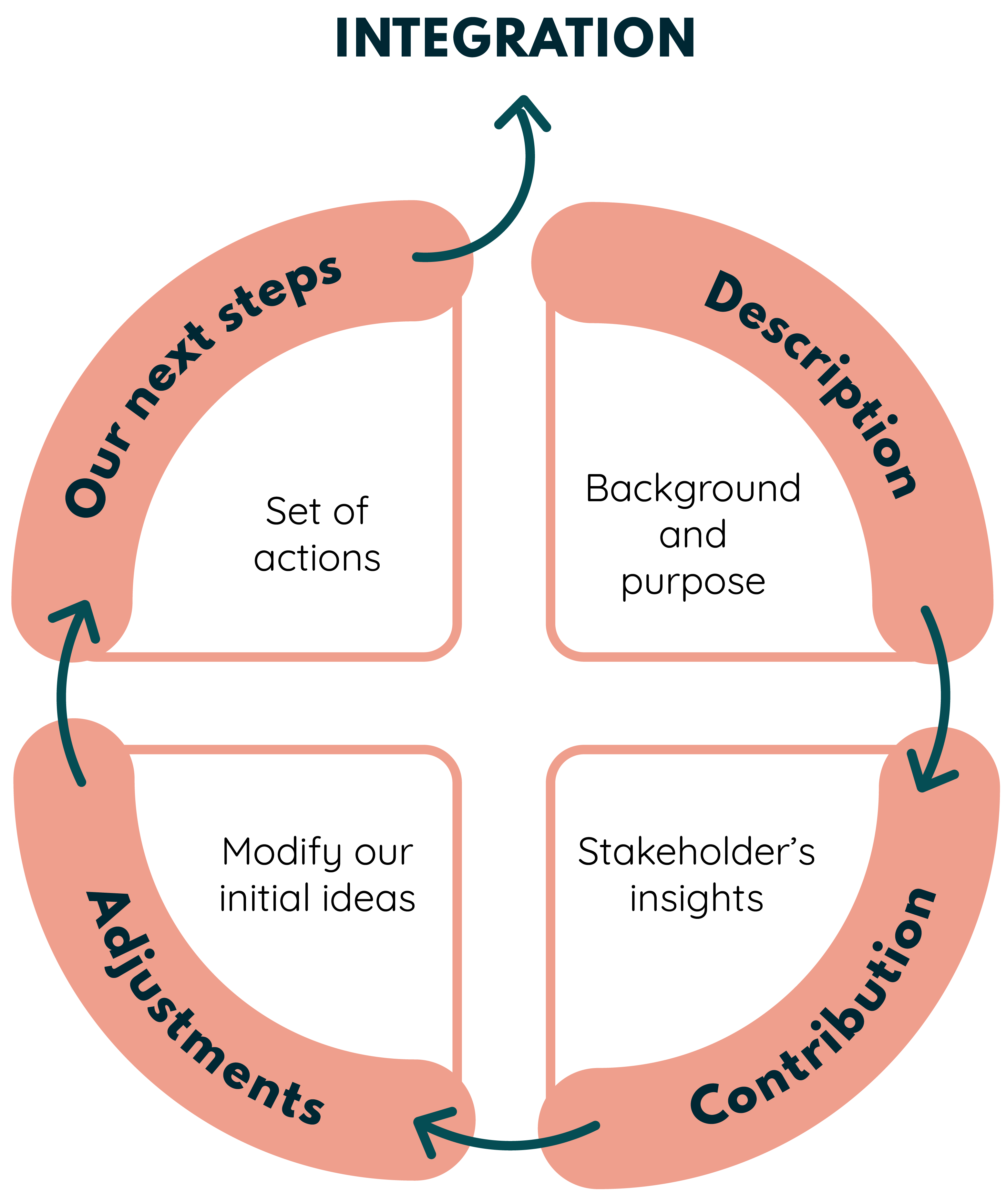
For structured and deliberate reflection of the consultations, we planned all of the Integrated Human Practices activities in one document accessible to all team members. Each stakeholder would have their line and instantly after the consultation, we would fill in the new information and update our upcoming tasks. The documentation consisted of four main parts, which emerged from RRI, AREA [3] frameworks, Exeter 2019, Vilnius-Lithuania 2020 Integrated human practices examples, and our vision of what are we expecting from the meetings.
Description
We briefly describe the background of the stakeholder, the purpose, and the course of the talk. To arrive at this point, we previously had to consider what answers we have to the questions raised, how we see
the possible solution to the design problem, impact on society.
Contribution
We understand this as hearing and considering the stakeholder’s view and insights on our suggested solutions or questions.
Adjustments
After learning the feedback from the stakeholder we then modify our initial ideas. We describe what insights we have accepted and tried to implement and which insights we have rejected.
Our next steps
In order for adjustments to be implemented, we need a set of actions, to begin with. This part shows where we begin the integration of the stakeholder’s insight into our project. In some of the meetings, we have outlined
explanations in italics for unfinished actions about why we have not succeeded in executing them.
Each of the consultations is described below in a timeline. Not every meeting had a follow-up and not all of what was planned was executed. This is mostly due to the limited time we have in the iGEM cycle, and the unresponsiveness of potential partners.
Even though until now only practices regarding amebiasis were described, our team has thoroughly worked with education and inclusivity activities, which were related not only to “AmeBye” but to synthetic biology as an area of science. Therefore we had consultations and stakeholders who have not had an impact on the wet or dry lab. However, they were significant to quality education and inclusive activities. We applied simple principles for these consultations: whenever we felt like we lacked experience in the topic or had questions, we reviewed the activity with a person from that particular field.
Conclusion and main changes
To conclude, in the wet lab processes, we made such stakeholder insights-based decisions: qualitative (not quantitative) test, prevention of intestinal amebiasis (not treatment of amebic liver abscess). User-friendly, low-cost disassemblable and durable test design along with educational information in both the test and the probiotics packaging was bolstered by stakeholders, market assessment, and social sciences research.
The planned dry lab consultations provided valuable insight into the fusion protein modeling area: we included more scorings of aptamer-protein complexes and introduced multiple sequence alignment into our flow. Although, since AlphaFold2 became available in the middle of our modeling cycle, the stakeholders of academia suggested we try the tool for our system. It was a major change in the flow since molecular dynamics simulations were no longer strictly required.
However, for further improvement, we see that it is essential to get to talk with travelers and people living in endemic regions or who have experienced amebiasis by themselves since they are considered as target users of the “AmeBye” tools.
Even though there is space for further development and improvement of our human practices, we put a lot of thought and effort into trying to perceive the impact of our project, surrounding context to arrive at evidence-based decisions relevant to the world. Our Human Practices significantly influenced our final project design and implementation.
References

